Atoms, Molecules, and Compounds
Key Terms:
|
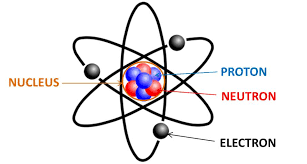
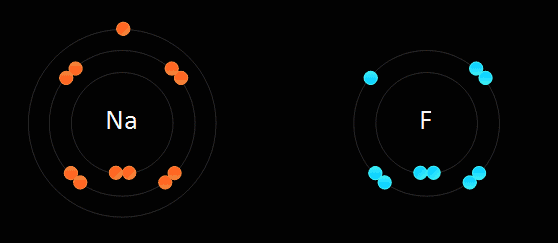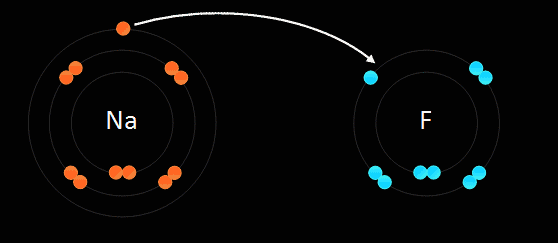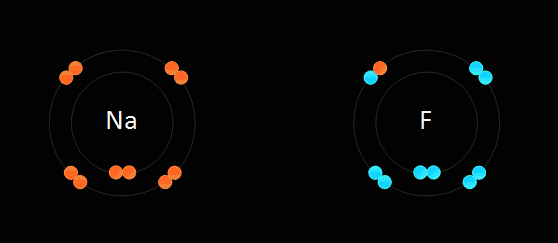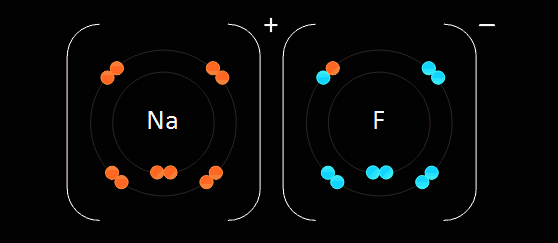
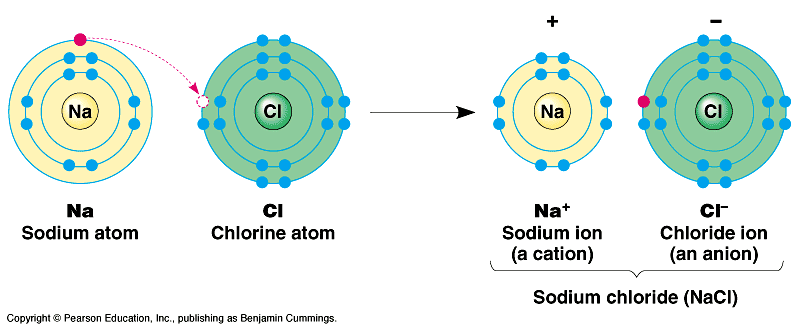
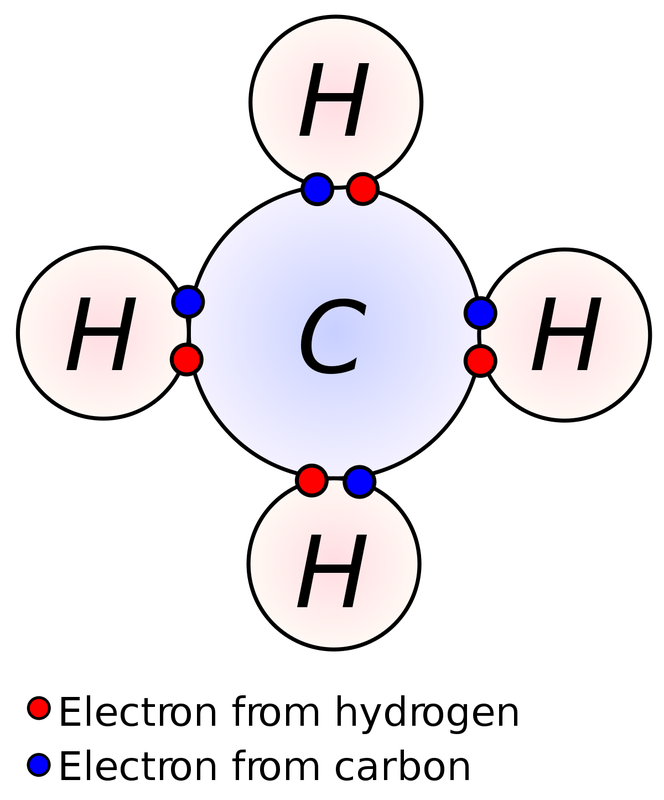
Atom - smallest unit of matter that still retains the properties of an element
|
Compound - 2 or more elements in a fixed ratio
|
Water
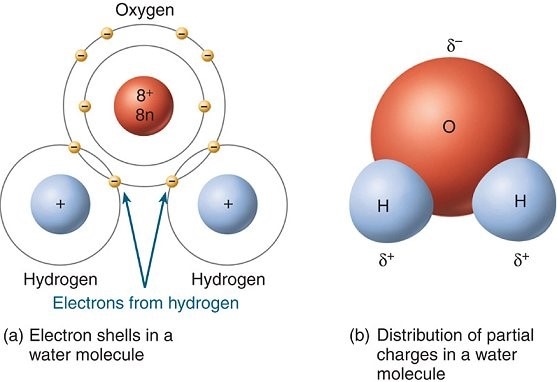
Water is a simple compound composed of 2 hydrogen atoms bonded with 1 oxygen. Although simple, water has unique characteristics that make it essential. Water's characteristics, or properties, are a result of water's polarity. Polarity is the unequal distribution of positive and negative charges between the covalent bonds of atoms in a compound.
Properties of Water
CohesionWater molecules are attracted to each other's polarity therefore water will create strong hydrogen bonds with other water molecules.
Surface Tension - how difficult it is to break the surface of a liquid. Water’s high surface tension is created by the cohesion of water. High Specific HeatEvery substance has a specific heat (the amount of energy the substance can absorb and retain)
Water has a high specific heat. Water takes a long time to absorb heat energy, but will retain the heat and slowly release the heat. Vineyards are usually around large bodies of water because water will slowly release heat into late fall extending the growing season.
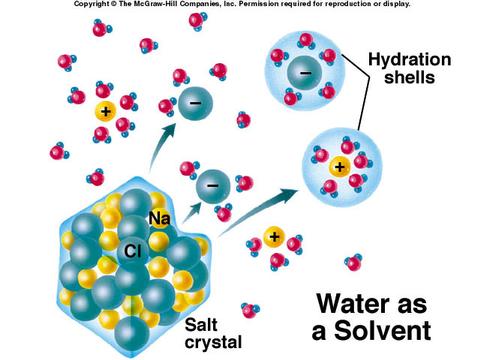
Universal SolventSolution – a mixture containing 2 or more substances.
Solute – the substance that is dissolved Solvent – the dissolving agent Water is able to dissolve MANY different types of solutes because water is polar! Water’s negative and positive charge will attract solutes to water. Water will bond and dissolve the solute. |
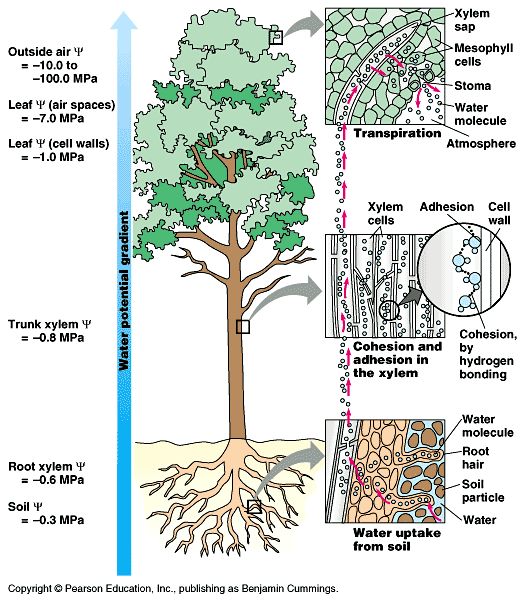
AdhesionAdhesion is the ability of water to bond or “cling” to other substances.
Capillary Action – plants use adhesion to pull water through the plants from the roots to the leaves. Water is most dense at 4 degrees.Water is most dense at 4°C
Water freezes at 0°C and will be less dense than cold water due to the spacing of hydrogen bonds between water molecules.… so ice floats! Evaporative CoolingWater cools a surface by evaporating.
Water absorbs high amounts of energy (high energy = heat) so when water evaporates and leaves the surface is left cool. |