What are Organic Compounds?
|
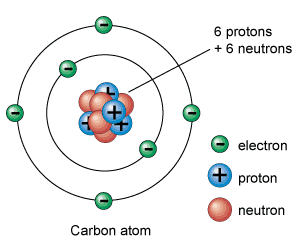
Organic Compounds are any CARBON - based molecules.
The element Carbon is the backbone of life because it has 4 electrons in its outermost electron shell. Carbon can make 4 covalent bonds because of its amount of electrons in its outermost electron shell. Hydrocarbons are compounds composed of only CARBON and HYDROGEN Isomers are compounds with the SAME molecular formula but DIFFERENT structural arrangement. |
Monomer vs. Polymer
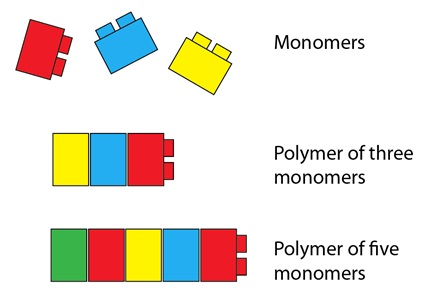
Monomers are small repeating sub units that make larger molecules called polymers. Monomers and polymers are just general names that can be used to describe any molecule. All large molecules we use on a daily basis are made up of smaller molecules. In each of the macromolecules we discuss in this unit have a specific monomer and polymers.
Dehydration Synthesis |
Hydrolysis |
|
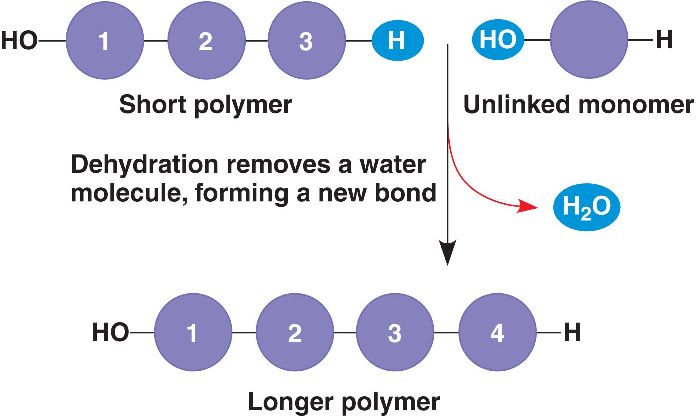
Dehydration Synthesis is the process of removing water to join molecules together.
|
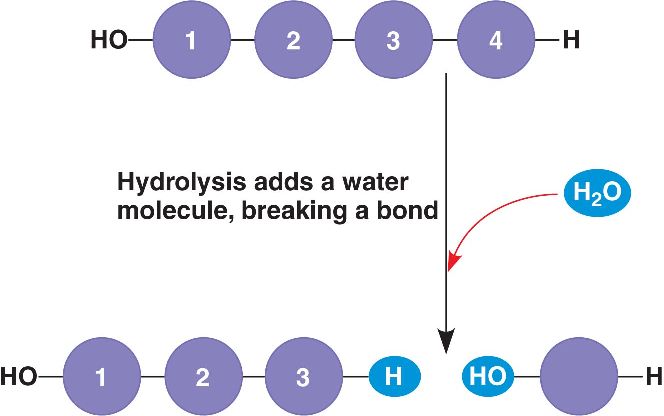
Hydrolysis is the process of adding water to break molecules down.
This is why we need saliva in our mouths! Saliva is water and enzymes that help break down the food we eat. |
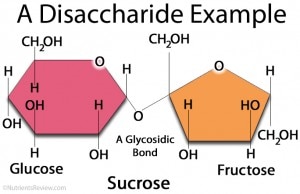
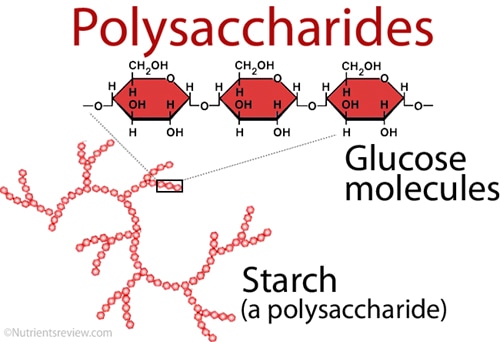
Carbohydrates
Carbohydrates are essential to life because carbohydrates are used in the production of energy.
Monomer: Monosaccharides |
Polymer: Disacchrides and Polysaccharides |
The Truth About Artificial Sweeteners
Read and view the articles and resources below to learn about artificial sweeteners. Use the articles and resources to formulate an educated opinion regarding artificial sweeteners and complete your activity provided in class.
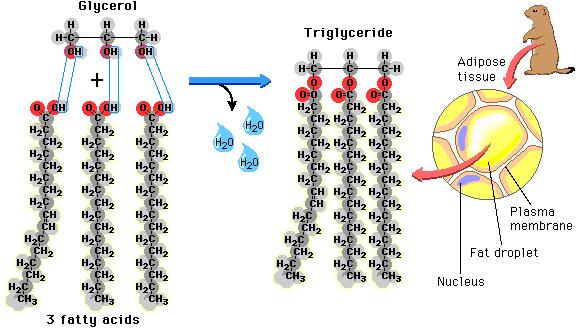
Lipids
Lipids have many different functions. Lipids are used for energy storage, membrane structures, and molecular signals. Lipids are unique because lipids do not have a true monomer, or repeating sub unit. Lipids are made of multiple components depending on the lipid's function.
Types of Lipids
TriglyceridesMade of 3 fatty acids and 1 glycerol
Used for energy storage |
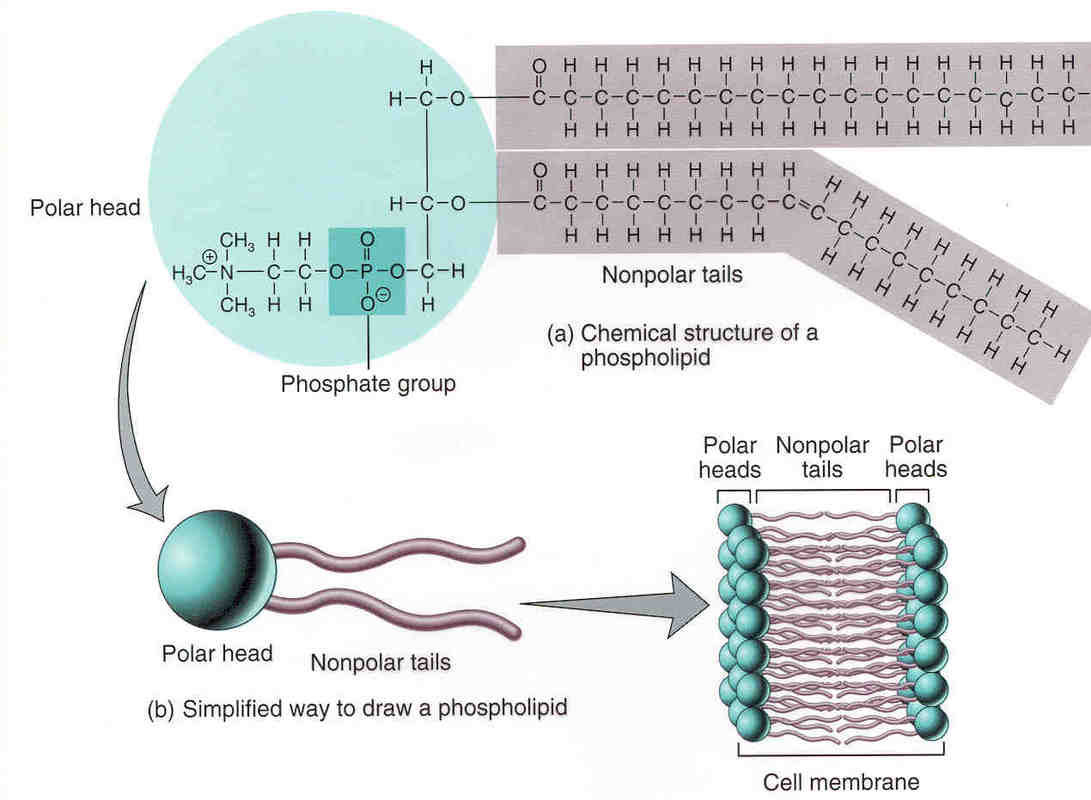
PhospholipidsMade of 1 phosphate group, 1 glycerol, and 2 fatty acids
Used in the cell membrane |
Saturated FatsA fat used in energy storage that contains ALL SINGLE BONDS in the fatty acid tails. The fatty acid is SATURATED with hydrogens
Saturated fats are derived from animal fats and are solid at room temperature. |
UnsaturatedA fat used in energy storage that contains ATLEAST ONE DOUBLE BOND in the fatty acid tails.
Unsaturated fats are derived from plant oils and are liquid at room temperature. |
WaxesWaxes are lipids with an alcohol or hydroxyl group attached to the fatty acid. Waxes are used commercially and in nature to waterproof.
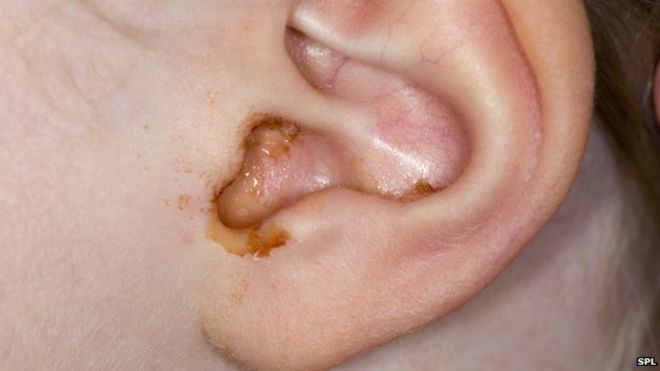
We produce earwax in our inner ear to create a protective waterproof layer. Earwax keeps fluids and small critters out of your ear!
This duck is not scratching himself! This duck is preening. Ducks and other water birds have a preen gland that secretes a wax. The wax is used to coat and waterproof ducks' feathers.
|
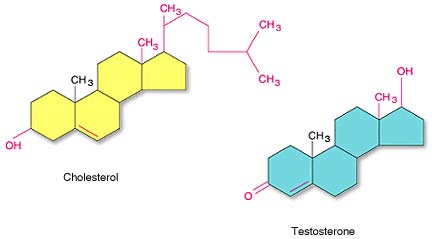
SteroidsSteroids are lipids containing 4-fused carbon rings. Steroids are used for many things. The steroid cholesterol is used to cell membrane structure while testosterone is the male sex hormone.
Not all steroids cause this... |
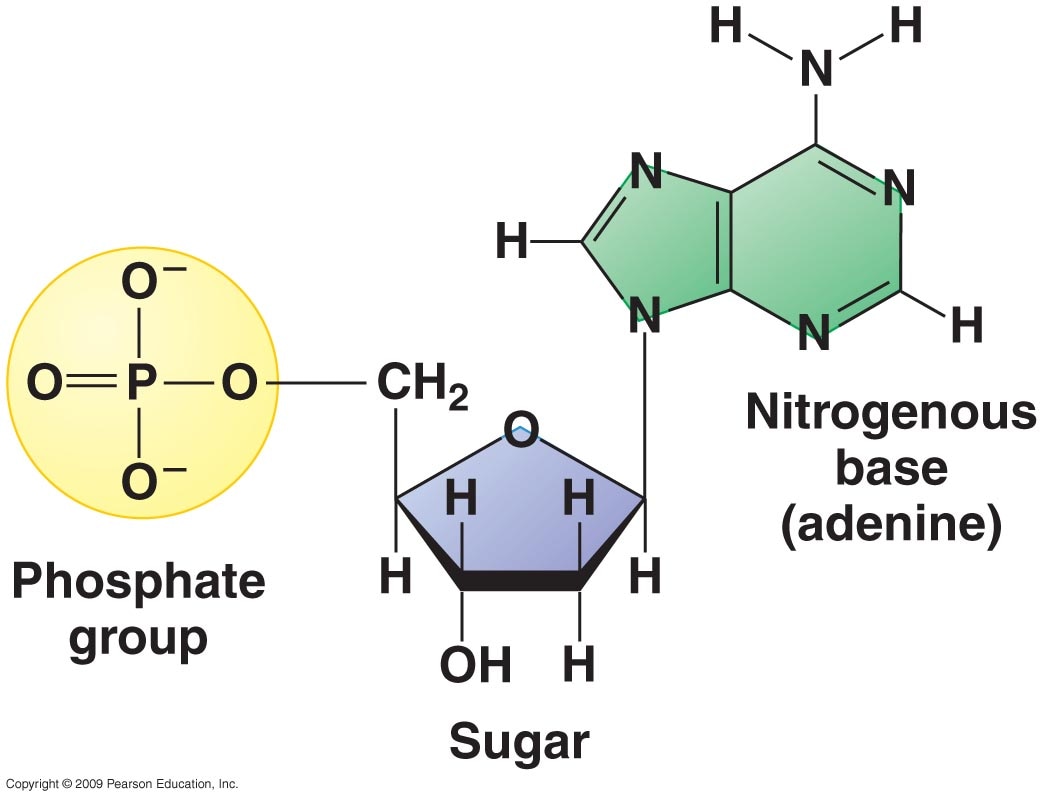
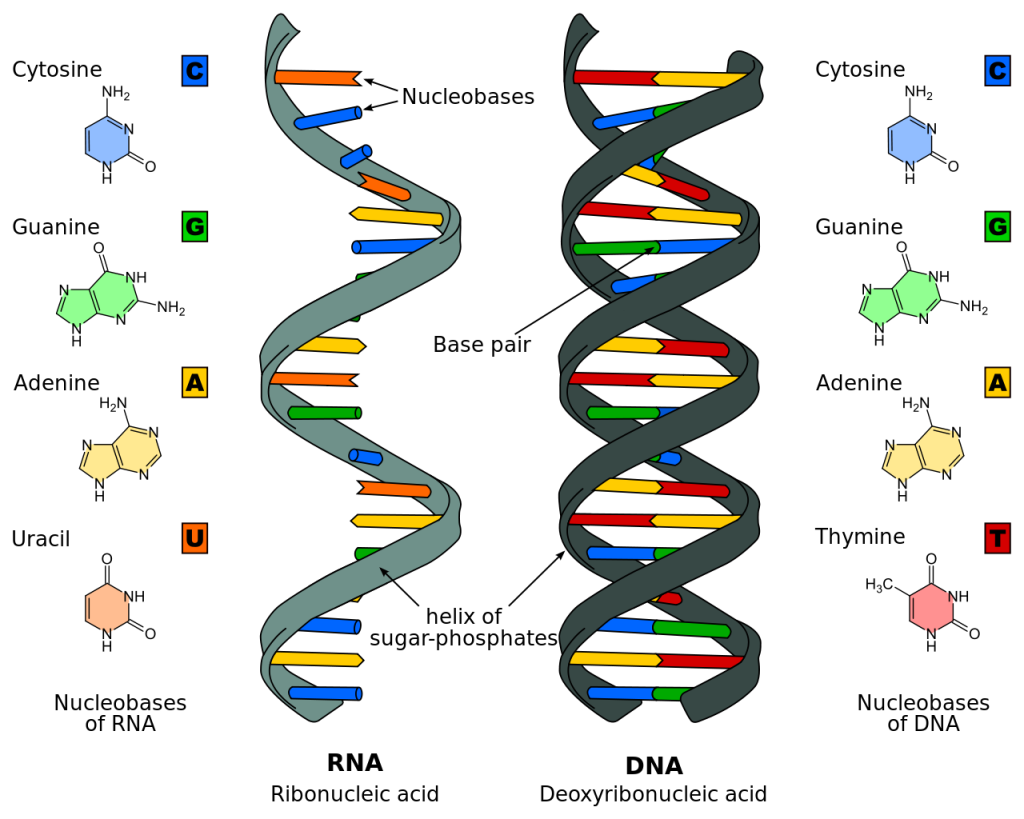
Nucleic Acids
Nucleic acids are the primarily used as the genetic material in each of our cells. More information about nucleic acids can be found in the "DNA to Protein Synthesis" unit.
Proteins
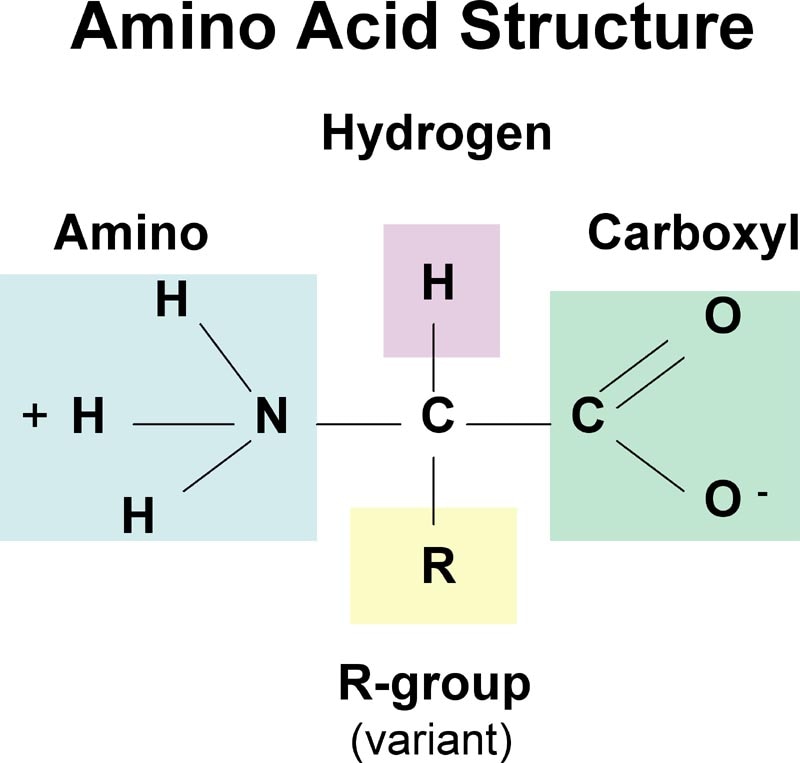
Monomer: Amino AcidThere are 20 amino acids. Each amino acid differs from the next because of their specific "R" group.
|
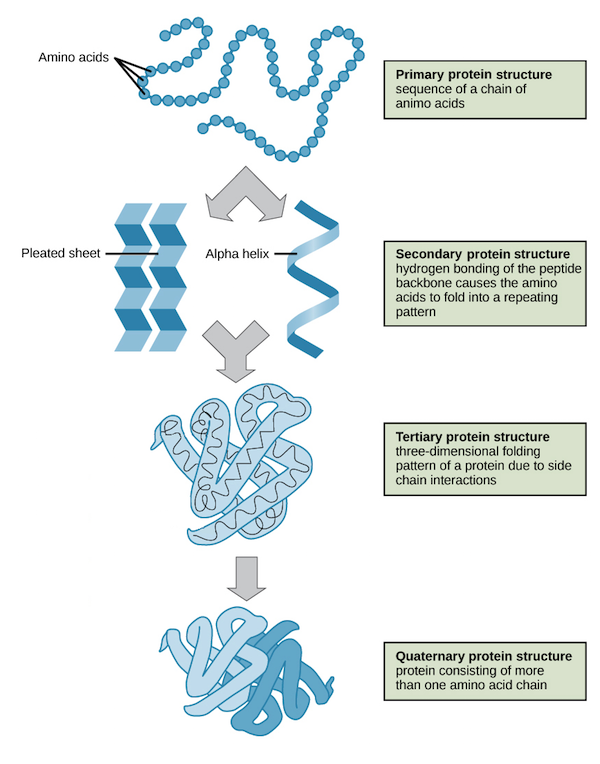
Polymer: PolypeptidePolypeptides possess a specific shape that results in a specific function. The specific shape occurs based on the chain of amino acids. Amino acids act as letters in an alphabet because the different combinations of letters create different words with unique meanings.
|
Denaturing
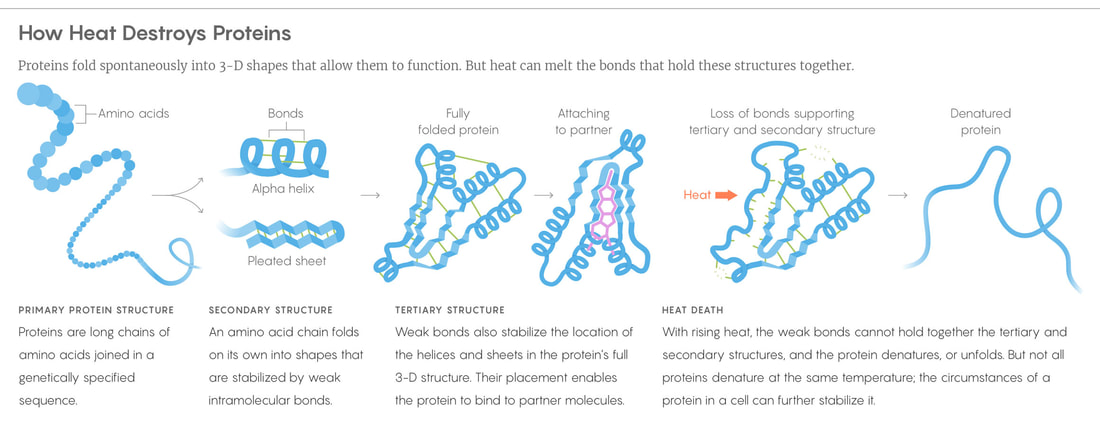
Denature – when a protein’s shape is changed by the following environmental changes:
- change in temperature
- change in pH
- change in salinity (salt concentration)
When a protein is denatured, the tertiary structure of the protein will unravel down to the primary structure (chain of amino acids) because the hydrogen and ionic bonds holding the 3D shape together break in extreme environmental changes.
- change in temperature
- change in pH
- change in salinity (salt concentration)
When a protein is denatured, the tertiary structure of the protein will unravel down to the primary structure (chain of amino acids) because the hydrogen and ionic bonds holding the 3D shape together break in extreme environmental changes.